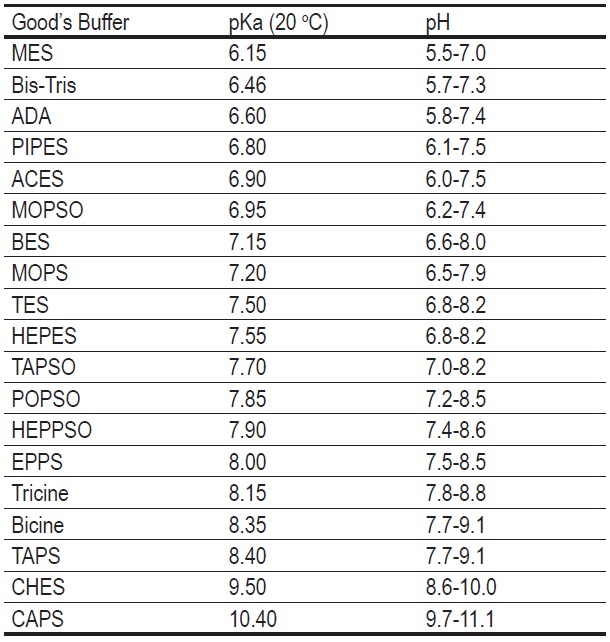
pH buffers that have been used with gametes and embryos from various... | Download Scientific Diagram

Solution pKa values for a range of polymers at various polymer and salt... | Download Scientific Diagram

SOLVED: You are trying to create a buffer with pH = 7.7. a.) Which acid from the table below should you use? b.) What else do you need to add to create

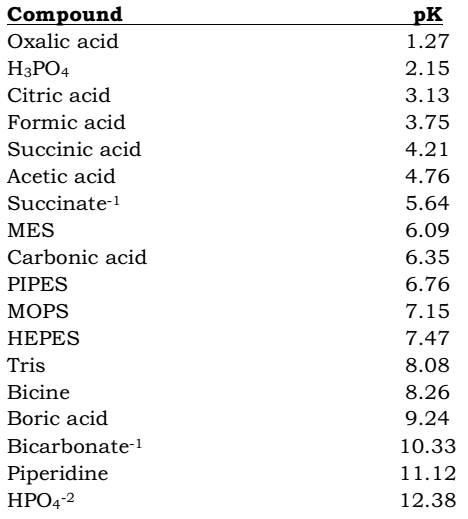
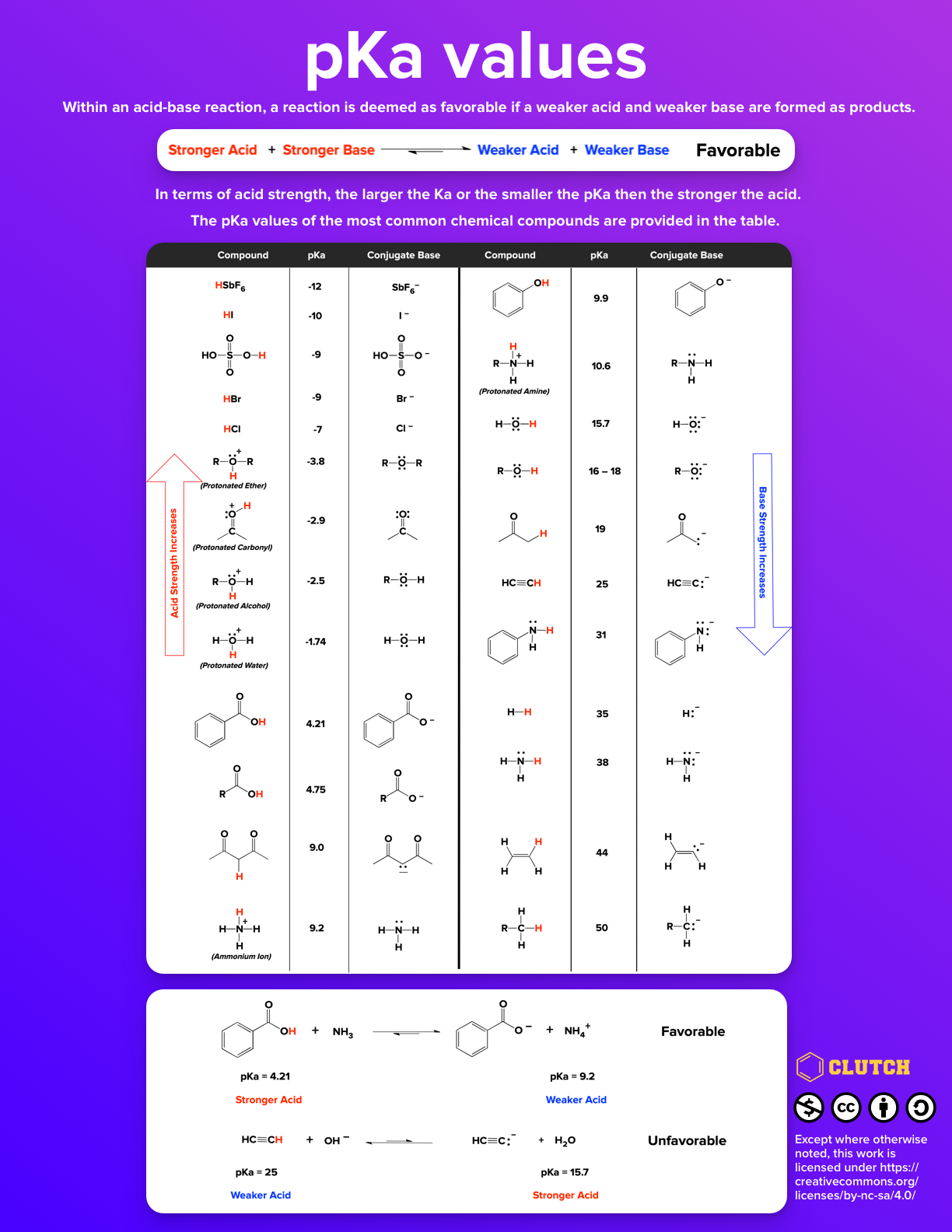
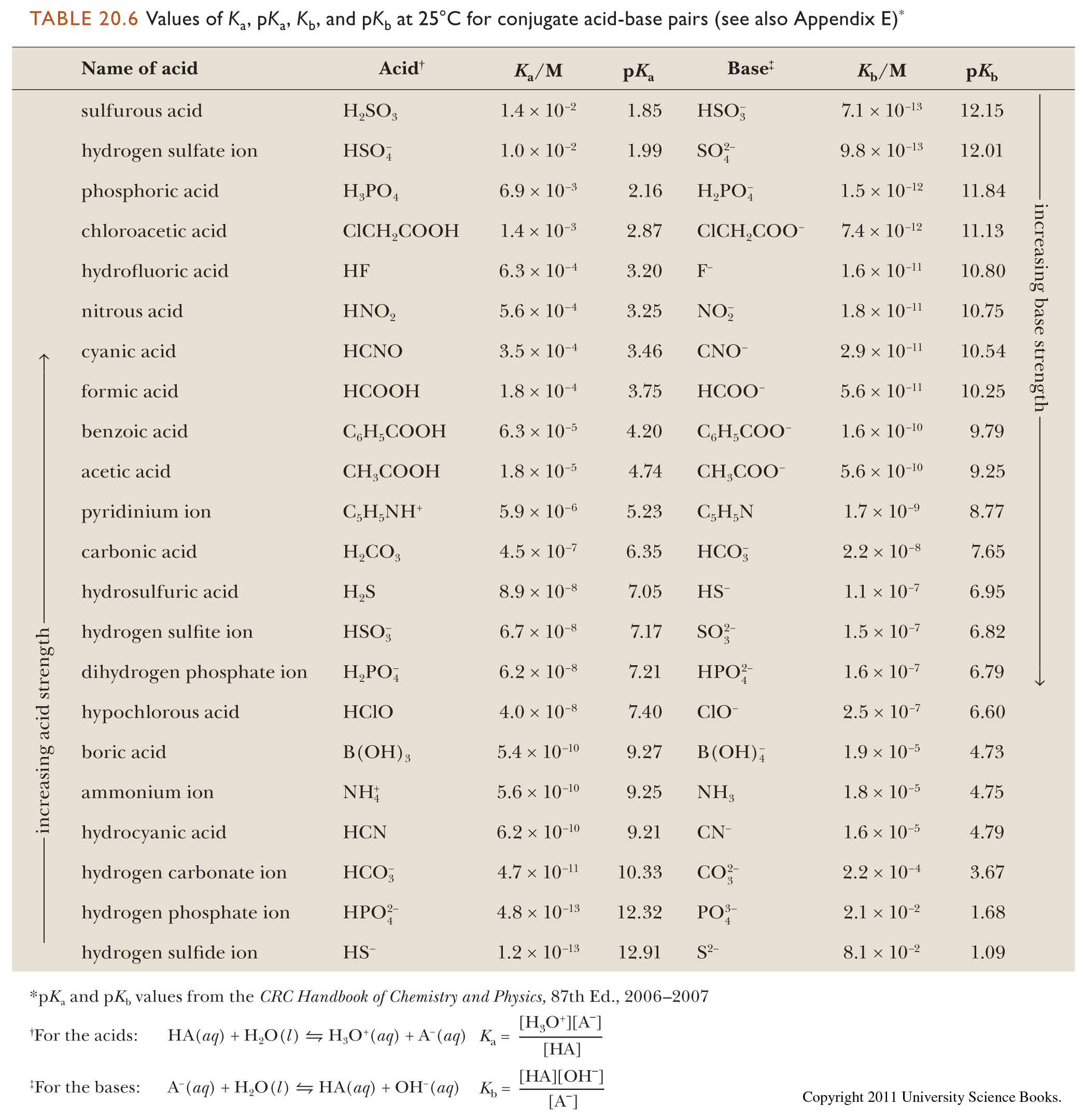
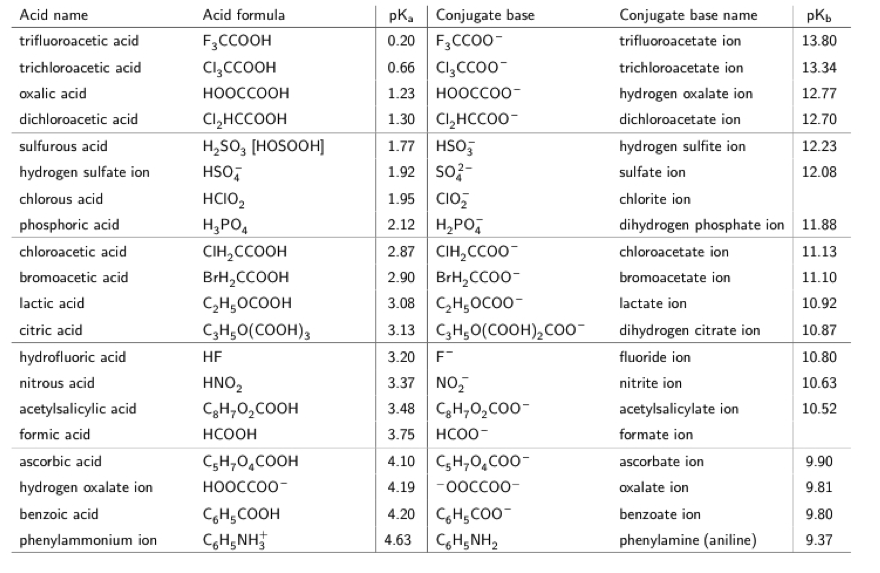
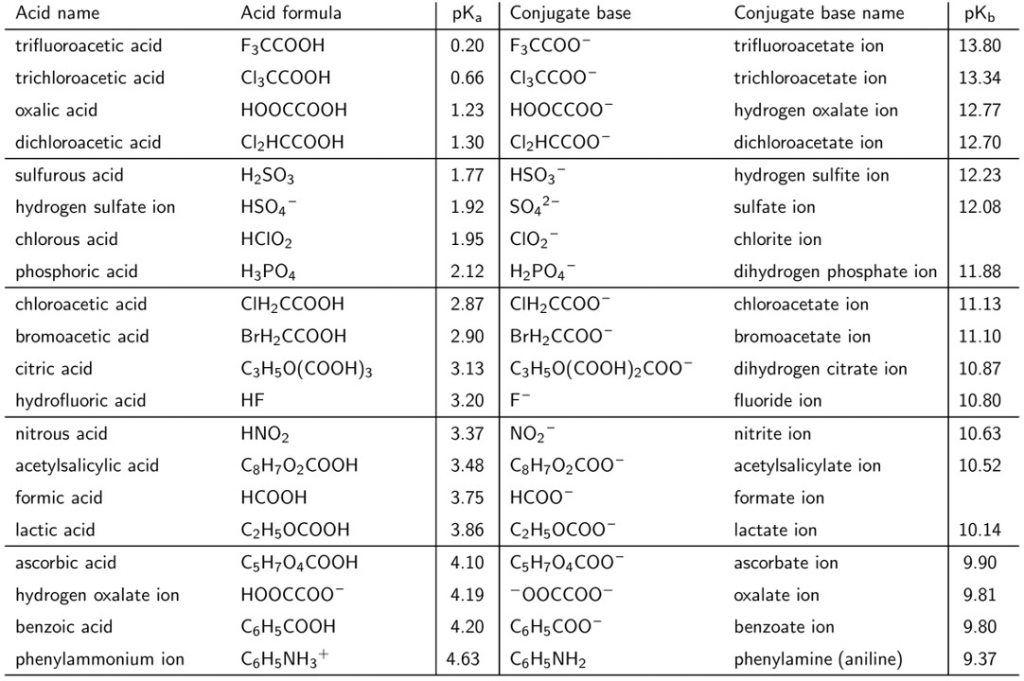
SOLVED: 11. Buffer preparation Given the KalpKa of the following compounds: TABLE 8.3 Some K; and pK; Values for Weak Acids, in Decreasing Order of Strength Name Formula K pK. Phosphorie acid
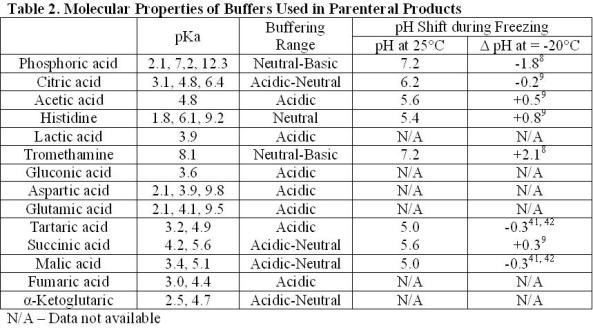
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review
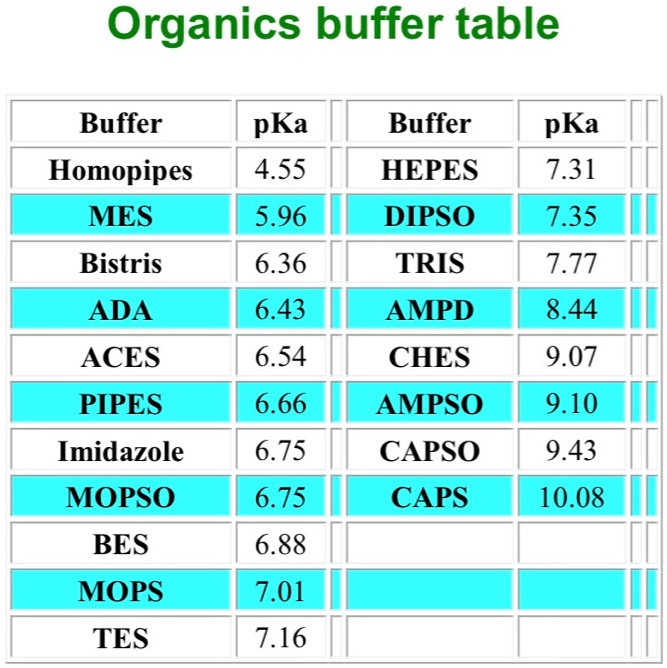
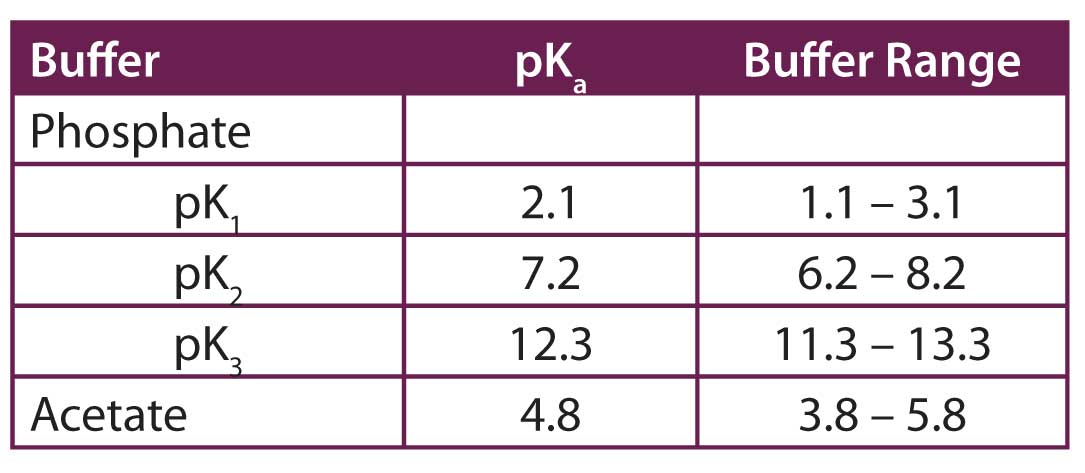
SOLVED: Organics buffer table Buffer Buffer Homopipes MES pKa 4.55 5.96 HEPES pKa 7.31 7.35 DIPSO Bistris pKa 6.36 TRIS AMPD pKa 7.77 ADA pKa 6.43 8.44 ACES PIPES pKa 6.54 CHES

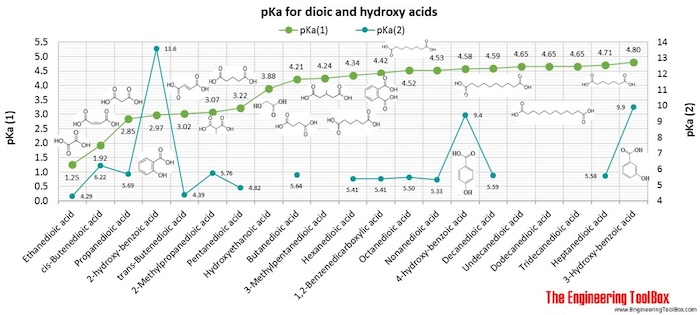
Table 4 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar

equilibrium - Why do buffers need to be composed of equal amounts of the acid and salt? - Chemistry Stack Exchange

Table 3 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar

How To Calculate the PH of a Buffer Solution | Equation & Example - Video & Lesson Transcript | Study.com

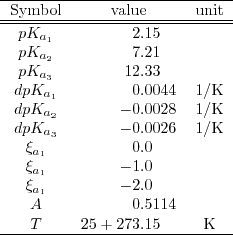
Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength - ScienceDirect
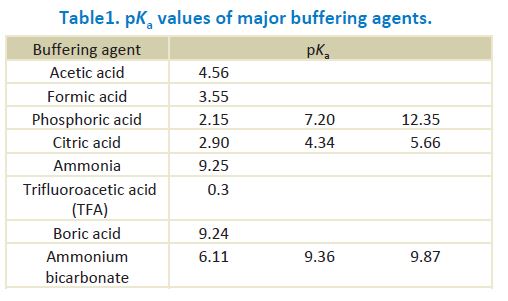
![PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0a6140f88db06398f56b28ce5fd6ac92cd82d39/5-Table3-1.png)